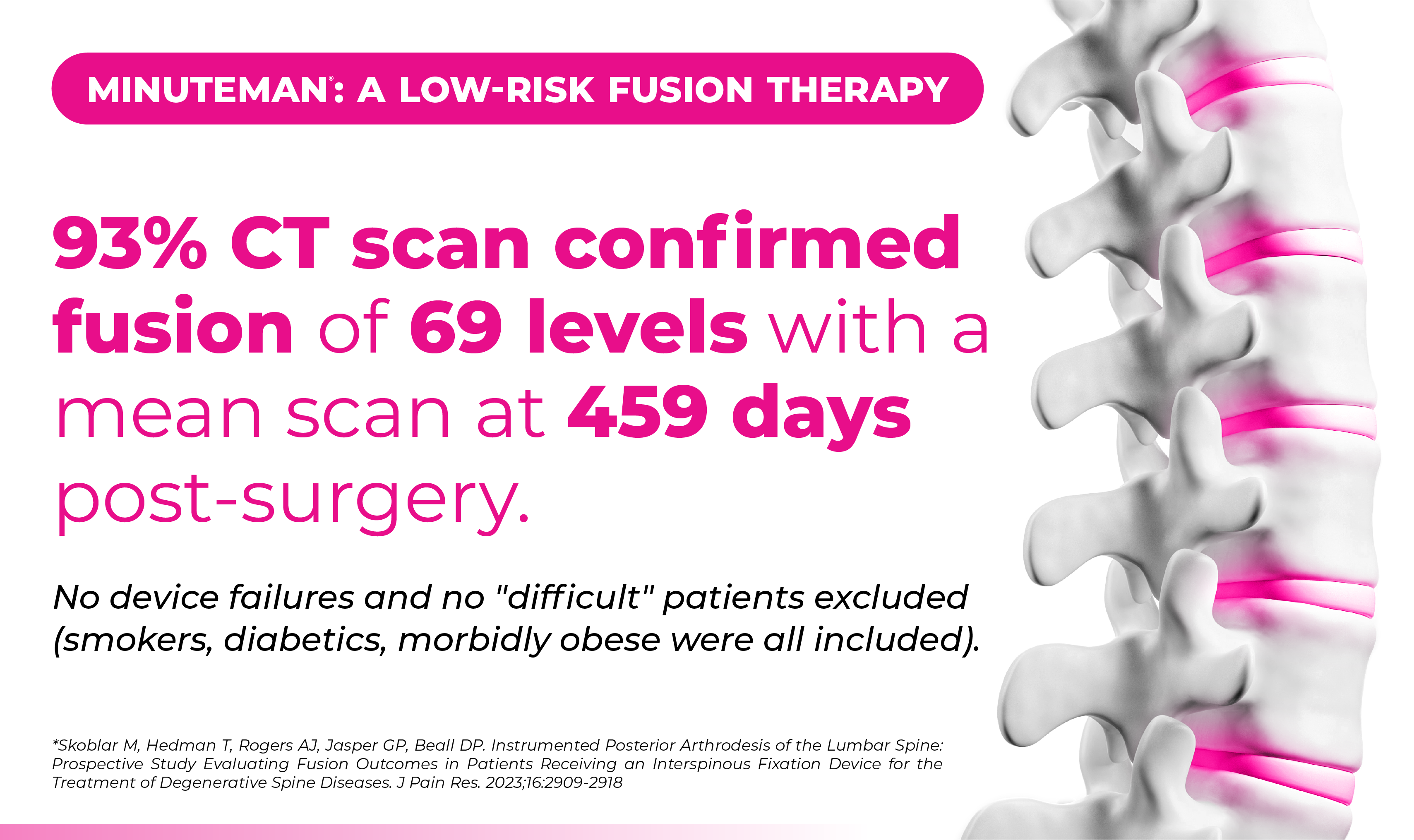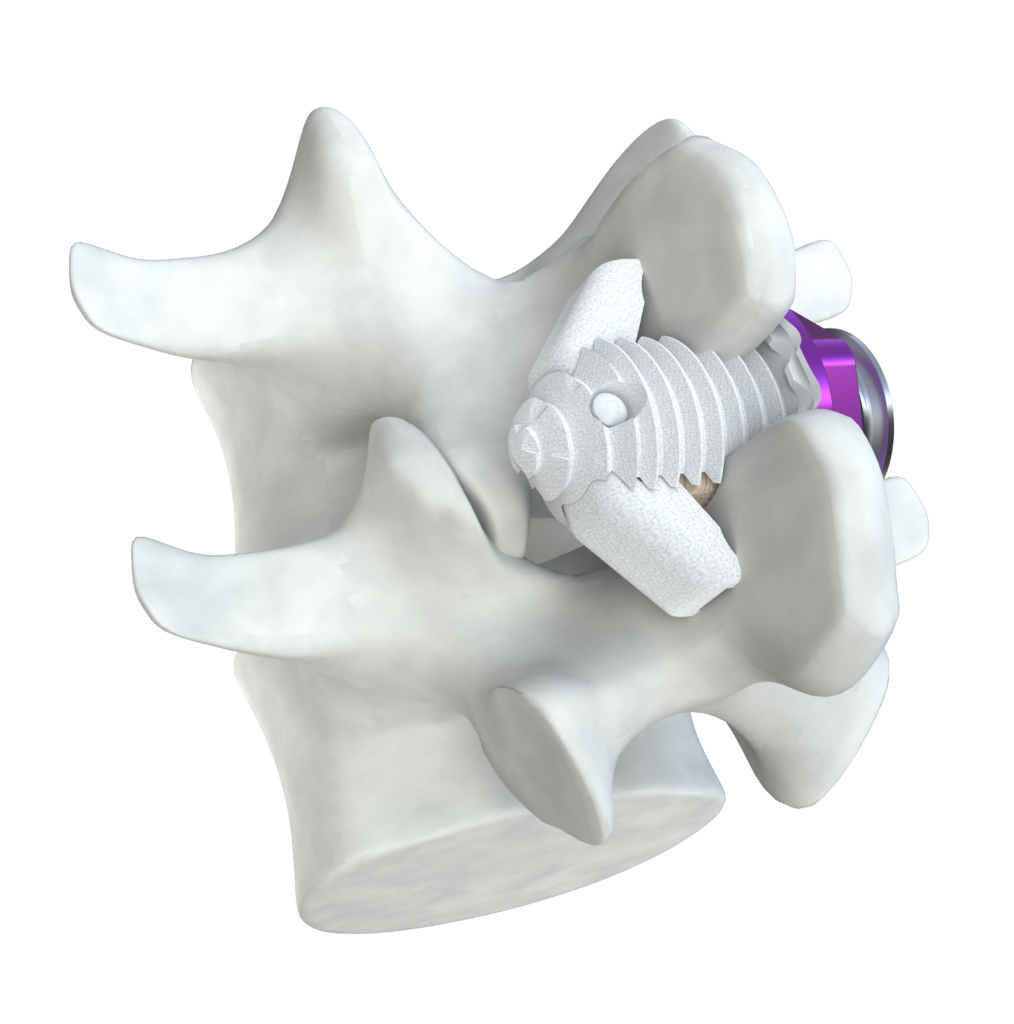

Spinal Simplicity’s Minuteman® is an interspinous-interlaminar fusion device. It is intended for the stabilization and fusion of the thoracic, lumbar, and sacral spine. The device is designed for attachment to the posterior non-cervical spine at the spinous processes through its bilateral fixation plates. The Minuteman® is intended for use with bone graft material placed within the device.
The Minuteman® provides immobilization, stabilization, and fusion of the spinal segments. The core threaded post allows for optimal placement and a wide range of sizes allows for enhanced anatomical fit.
Conditions Treated
It is important you talk to your doctor about the best way to treat your back and/or leg pain. Your doctor will decide if the Minuteman® implant is the right choice for your condition.
Scientific Publications
Spinal Simplicity is dedicated to advancing spine care through the dissemination of high-quality research. Below, you will find publications that focus on the technology and its impact on the field. To access the publications, simply click on the images below.
Ready to revolutionize healthcare in Western Pennsylvania? Spinal Simplicity is seeking a dedicated addition to our team! For more information, connect with Eric Roux at eroux@spinalsimplicity.com and let`s drive change together!
#NowRecruiting #NowHiring #JoinOurTeam #SpinalSimplicity #TeamMinuteman #TeamLiberty

A great experience at the FSIPP & FSPMR`s Hands-On Lab in Orlando, FL!
A big thank you to Dr. Jonathan Daitch for his excellent instruction on Posterolateral Arthrodesis with Minuteman G5. Together, we`re making strides in spinal care! #FSIPP #FSPMR
#SpinalSimplicity #TeamMinuteman #ThinkAi

Join our team! Spinal Simplicity is on the lookout for a driven Sales Rep to join our team in Indiana. Interested in making an impact? Reach out to Tom Frazer at tfrazer@spinalsimplicity.com for more details.
#NowRecruiting #NowHiring #JoinOurTeam #SpinalSimplicity #TeamMinuteman #TeamLiberty

Set against the picturesque backdrop of Lake Oconee, GSIPP 2024 was a weekend of invaluable collaboration among physicians, midlevel practitioners, nurses, and industry peers, all dedicated to advancing interventional pain practice.
A heartfelt thank you to Drs. Nok Keomahathai, Chris Bovinet, Rutvij Shah, Joe Matthews, Travis Bailey, and Jordan Tate for inspiring connections and shared expertise, shaping the future of pain management together. #GSIPP24
#SpinalSimplicity #TeamMinuteman #TeamLiberty #Minuteman #LibertySI

Join us in shaping the future of healthcare in Washington DC! Spinal Simplicity is seeking a dedicated Sales Rep to join our team. For further details, contact Eric Roux at eroux@spinalsimplicity.com. Let`s make a difference together!
#NowRecruiting #NowHiring #JoinOurTeam #SpinalSimplicity #TeamMinuteman #TeamLiberty

Spinal Simplicity is proud to support FSIPP & FSPMR at their Hands-On Lab in Orlando, FL this weekend! We are privileged to have Dr. Jonathan Daitch teach Posterolateral Arthrodesis utilizing the Minuteman G5. #FSIPP #FSPMR
#SpinalSimplicity #TeamMinuteman #ThinkAi

Spinal Simplicity is expanding into Michigan, and we want you to join our team! We`re seeking a driven Sales Rep to help drive our growth. For more information, please reach out to Tom Frazer at tfrazer@spinalsimplicity.com.
#NowRecruiting #NowHiring #JoinOurTeam #SpinalSimplicity #TeamMinuteman #TeamLiberty

Spinal Simplicity is honored to exhibit at GSIPP this weekend! Come see Cole Davis and TJ Jackson for a live demo of Minuteman and Liberty-SI. #GSIPP24
#SpinalSimplicity #TeamMinuteman #TeamLiberty

Join our team at Spinal Simplicity and be part of our growth in Southern California! We`re currently looking for a dedicated Sales Rep to join our team. For more details, contact Neil Dougherty at ndougherty@spinalsimplicity.com. Let`s elevate healthcare together.
#NowRecruiting #NowHiring #JoinOurTeam #SpinalSimplicity #TeamMinuteman #TeamLiberty

Excellent discussion at ASIPP about Minuteman eligible patients who may have Angular Instability with Dr. Do Chan and Dr. Boris Shwartzman.
#ThinkAi #TeamMinuteman